New Global Survey Sheds Light on Pervasive Obstacles to Commercial Efficiency
Nearly two-thirds of respondents unable to distribute content across channels, yet 78% say it’s crucial
PLEASANTON, CA — Feb. 10, 2016 — Released today, the full findings from Veeva 2015 Life Sciences Commercial Content Management Survey reveal the industry’s patchwork approach to commercial content management is having a substantial impact on compliance and speed to market. The global survey of more than 250 commercial leaders shows striking deficiencies across the digital supply chain – from content creation and review to expiration, distribution, and withdrawal.
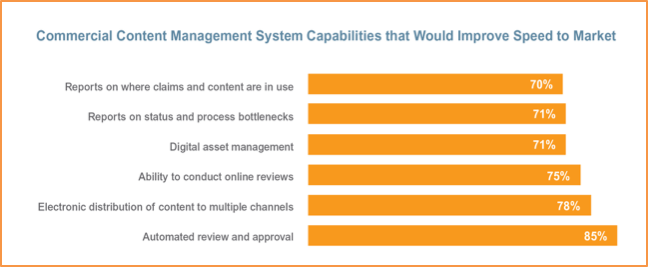
Despite the proliferation of digital channels, the majority (60%) of respondents to the industrywide survey cannot electronically distribute approved commercial content across multiple channels though most (78%) see it as crucial to improving time to market. Withdrawal of content is also a challenge, as nearly half (48%) of respondents still manually handle withdrawal.
The research also found many respondents (70%) lack a centralized, global digital asset management system, which can result in slower time to market and increased commercial content management costs. Additionally, 83% of respondents share commercial content with stakeholders via inefficient methods. For example, more than half rely on email to complete at least one or more tasks such as review and approval and content distribution, while 41% still use paper-based processes for certain functions.
These commercial content management shortcomings are exacerbated by increased volumes of content, additional communications channels, and a larger, more diverse group of stakeholders. The result is progressively longer content development cycles.
“The industry is churning out content at an incredible pace today, plus each asset is often tailored to individuals and produced in multiple formats to be leveraged across channels,” said John Chinnici, vice president, Veeva Vault PromoMats. “Life sciences companies cannot continue to rely on a patchwork of systems and paper-based processes in this new, highly complex commercial paradigm and expect to meet efficiency, timeline, and sales goals.”
The full survey follows the preliminary report released in October 2015, which revealed deficiencies impacting compliance. The vast majority (81%) of respondents, for instance, cannot report on where claims and content are in use. Almost half (49%) do not have primary systems that provide a full audit trail to manage commercial content throughout its lifecycle, yet most report that this capability would improve compliance.
Results of the annual study demonstrate the urgent need for a single commercial content management solution that provides complete visibility across the digital supply chain in order to keep up with the rapidly changing industry. Veeva Vault PromoMats provides a central, globally accessible digital asset management system with full audit trail across the digital supply chain plus electronic review, distribution, and withdraw.
The Veeva 2015 Life Sciences Commercial Content Management Survey explores the industry’s progress in transitioning to automated methods of managing commercial content across the digital supply chain. Drawn from the experiences and opinions of more than 250 regulatory, marketing, and medical leaders worldwide, the goal of this research is to understand the key capabilities required to speed commercial content time to market, and maintain optimal compliance.
Additional Information
For more on Veeva Vault PromoMats, visit: veeva.com/PromoMats
Stay updated on the latest Veeva news on LinkedIn: linkedin.com/company/veeva-systems
Follow @veevasystems on Twitter: twitter.com/veevasystems
Like Veeva on Facebook: facebook.com/veevasystems
About Veeva Systems
Veeva Systems Inc. is a leader in cloud-based software for the global life sciences industry. Committed to innovation, product excellence, and customer success, Veeva has more than 375 customers, ranging from the world's largest pharmaceutical companies to emerging biotechs. Veeva is headquartered in the San Francisco Bay Area, with offices in Europe, Asia, and Latin America. For more information, visit www.veeva.com.
Forward-looking Statements
This release contains forward-looking statements, including the market demand for and acceptance of Veeva’s products and services, the results from use of Veeva’s products and services, and general business conditions, particularly in the life sciences industry. Any forward-looking statements contained in this press release are based upon Veeva’s historical performance and its current plans, estimates, and expectations, and are not a representation that such plans, estimates, or expectations will be achieved. These forward-looking statements represent Veeva’s expectations as of the date of this press announcement. Subsequent events may cause these expectations to change, and Veeva disclaims any obligation to update the forward-looking statements in the future. These forward-looking statements are subject to known and unknown risks and uncertainties that may cause actual results to differ materially. Additional risks and uncertainties that could affect Veeva’s financial results are included under the captions, “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in the company’s filing on Form 10-Q for the period ended October 31, 2015. This is available on the company’s website at http://www.veeva.com under the Investors section and on the SEC’s website at www.sec.gov. Further information on potential risks that could affect actual results will be included in other filings Veeva makes with the SEC from time to time.
###
Contact:
Public Relations
Veeva Systems Inc.
pr@veeva.com