
Smith+Nephew drives faster document creation and 90% time savings in quality control with Veeva eTMF.
View Customer Story

Gore standardizes processes, reduces duplicate data entry, and better enables sites
View Customer Story

Terumo unifies global clinical trials to ensure standardization and collaboration with Veea eTMF and Veeva CTMS
View Customer Story
Drive efficient and compliant trials
Veeva eTMF is the leading trial master file application to ensure the quality, timeliness, and completeness of a TMF. It provides full enterprise content management capabilities for upload, version control, QC and approval, and real-time co-authoring with Microsoft Office for study documents such as consent forms. It’s highly efficient and performant and supports global outsourcing.
Completeness and timeliness are managed through Expected Document Lists (EDLs). Content files are auto-classified through the TMF Bot, and classified content is matched automatically to EDLs. The TMF Transfer feature simplifies exchange between sponsors and CROs by sending completed trial master files at study close.
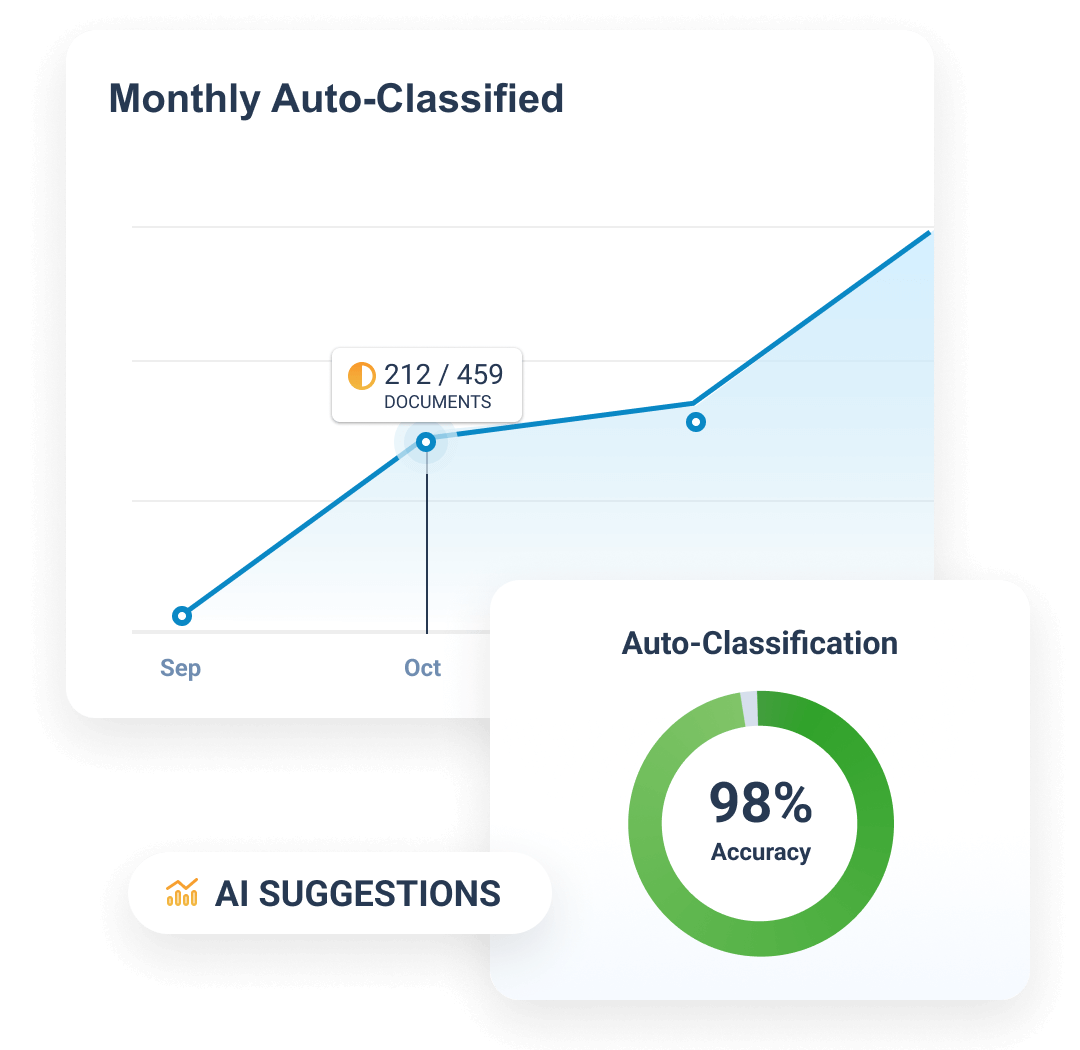
>98%
auto-classification accuracy with TMF Bot
75%
faster TMF delivery in outsourced models
40%
cut in study reconciliation time
Veeva eTMF Impact
Stay inspection ready
Manage all TMF documents and processes in one system to ensure ongoing inspection readiness.Improve data quality
Streamline registration management by reducing data duplicates and discrepancies.Provide global visibility
Stay informed with a complete view into the marketing status of your global product portfolio.Speed health authority responses
Manage product registration queries and commitments to stay ahead of response deadlines.
Customer Success
Medtechs speed trials with Veeva Clinical Operations































Watch video
Smith+Nephew shares how Veeva eTMF drives faster document creation and 90% time savings for quality control

Watch video
Gore standardizes processes, reduces duplicate data entry, and better enables sites

Read article
Terumo unifies global clinical trials to ensure standardization and collaboration with Veea eTMF and CTMS

Download infographic
The MedTech Clinical Benchmark examines how companies are increasing trial activity to maintain product marketability

Watch video
Integra shares how unifying clinical data and clinical operations established repeatable processes data visibility

Watch video
Bio-Rad harmonizes global processses with Veeva eTMF to reduce discrepancies, inconsistencies, and compliance issues




